Understanding AI workflows are a vital part of created embedded edge applications.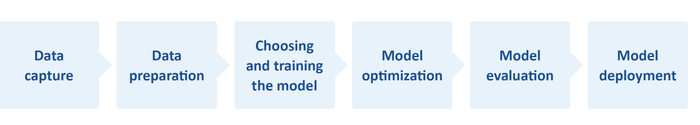
Embedded Systems
How Can Embedded Engineers Implement Edge AI ApplicationsHow Can Embedded Engineers Implement Edge AI Applications
Following these useful tips can help you create edge AI applications without straining processor and storage resources.
Sign up for the Design News Daily newsletter.





















.jpg?width=300&auto=webp&quality=80&disable=upscale)






















