New Self-Charging Battery Eyed for Electronic Devices
The cell, developed by collaborators in the U.S. and Portugal, combines negative capacitance and negative resistance to recharge without losing power.
April 2, 2020
One of the least user-friendly things about ubiquitous use of electronic devices such as cell phones and tablets is the need to charge the battery after prolonged use. A collaborative team of researchers based in Portugal and the United States may be on their way to solving this issue with the development of a new type of battery that can re-charge itself without losing energy.
|
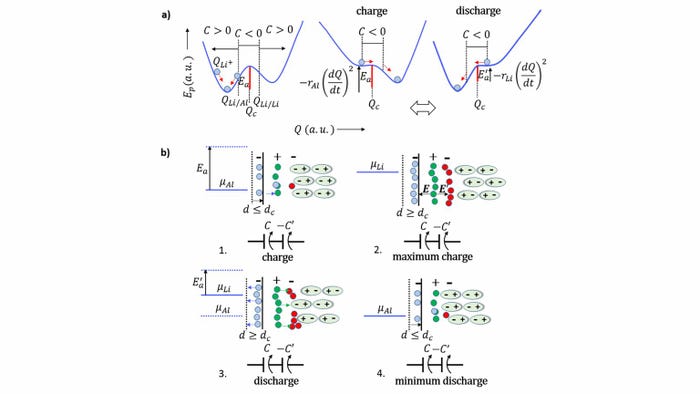
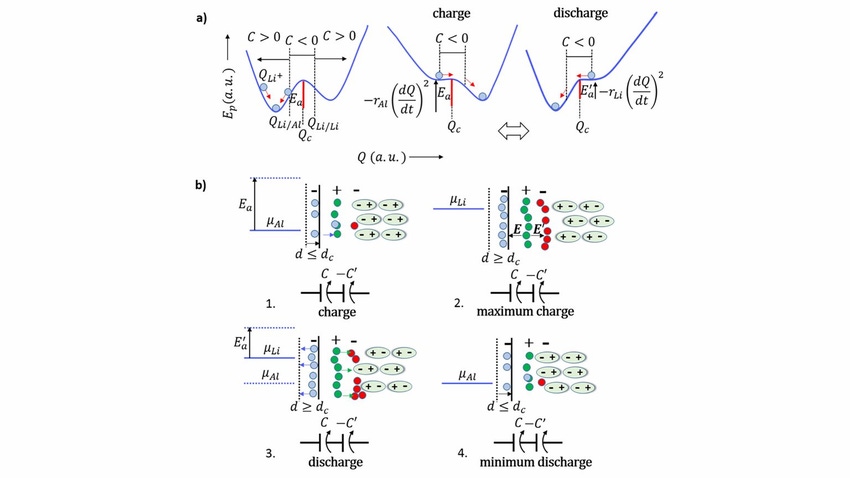
The design of the engineering behind a self-charging battery developed by researchers at the University of Porto in Portugal and the University of Texas at Austin. (Image source: Braga et al.) |
The device—developed by engineers at the University of Porto and the University of Texas at Austin—combines negative capacitance and negative resistance within the same cell to allow it to recharge without power loss.
Negative capacitance occurs when a decrease in voltage across a material or device leads to an increase in its electrical charge. Negative resistance is the opposite—it happens when an increase in voltage across a device or material results in a decrease in the electric current running through it.
Having these two co-exist in the same battery is significant because it unifies the theory behind all solid-state devices—which include not only batteries but also capacitors, photovoltaics, and transistors, said Helena Braga, an associate professor in the Engineering Physics Department at the University of Porto. The theory shows that different materials in electrical contact exhibit the properties of the combined material instead of those of the individual materials, said Braga, who led the research.
In a battery, this plays out in the open circuit potential difference between electrodes, which is due to an electrical need to align the Fermi levels, she said. These levels are a measure of the energy of the least tightly held electrons within a solid, something that also is responsible for the polarity of the electrodes, Braga said.
Aligning for the Charge
“When one of the materials is an insulator or dielectric, such as an electrolyte, it will locally change its composition to form capacitors that can store energy and align the Fermi levels within the device,” Braga explained in a press statement. “Our electrochemical cells, which in principle are simpler than batteries, are all about self-organization, which is the substance of life.”
Researchers developed a lithium-rich glass electrolyte for their battery, which can feed both electrons with lithium ions, Braga said. This means researchers didn’t have to use lithium metal in the design, she said. Indeed, they designed the battery using just two different metals as electrodes and the glass electrolyte—which could also be created using sodium--between them.
To create a more sustainable source of energy, researchers said the device’s self-cycling can be stopped or mitigated by not allowing a leap in the Fermi levels or by configuring a negative resistance to happen, Braga added.
“This can be obtained by having the negative electrode of the same material as the positive ions of the electrolyte,” she said in a press statement. “It gives rise to a device that self-charges without self-cycling--increasing the energy stored in it--as opposed to the natural degradation of the electrochemical process that makes the energy stored decrease by dissipation of heat.”
The approach researchers took can be applied to all energy-storage devices, such as batteries and capacitors, to help improve their autonomy and allow for a similar self-charging principle, Braga said.
While the technology her team developed is not strong enough to power a mobile device—it’s currently suited for use in in extremely low-frequency communications, such blinking lights, electronic beepers, inverters, or digital converters—one day it could scale for larger devices, she added.
RELATED ARTICLES:
Elizabeth Montalbano is a freelance writer who has written about technology and culture for more than 20 years. She has lived and worked as a professional journalist in Phoenix, San Francisco and New York City. In her free time she enjoys surfing, traveling, music, yoga and cooking. She currently resides in a village on the southwest coast of Portugal.
About the Author(s)
You May Also Like