Accelerating Medical Device Innovation Using Additive Manufacturing
November 5, 2015
Additive manufacturing opens new dimensions for integrated design, development, and production. AM is an important technological innovation that helps companies break existing performance tradeoffs in four fundamental ways:
First, it increases design, development and production flexibility and reduces the capital needed to achieve a variety of different design options.
Second, it enables more agile-like product development processes and accelerates the product development cycle.
Third, it allows companies to de-risk major programs, and approach bold new ideas with greater early stage risk-tolerance and late-stage confidence.
Fourth, AM helps reduces the capital involved in achieving economies of scale.
However, deciding when, where, and how AM can be used to allow more design freedom, compress the product development cycle time, and commercialize more innovative products is not trivial. Medical Device manufacturers interested in adopting AM for surgical equipment, prosthetics, implants, and tissue engineering need to consider many factors to determine if digital development and production technologies are appropriate and if so, where along the product development and manufacturing line they ought to be leveraged.
On the practical side, medical device manufacturers need to determine which materials (polymers, metals, ceramics, and other biocompatible materials) ought to be used for initial proof of product and for final production. They then need to consider the appropriate deposition technologies (Fused Deposition Modeling, Stereolithography, Selective Laser Sintering/Melting, Laminated Object Manufacturing, Electronic Beam Melting, or Inkjet Printing) that meet the design and manufacturing requirements. Companies in all industries need to consider product feature requirements, development effort, time to market, and production volume when they pursue new innovations. But in some industries, such as medical devices, there is a greater market need and emphasis placed on performance. Devices that can perform a particular procedure more effectively and efficiently are very valuable. There are many benefits to conventional manufacturing that cannot be matched by additive techniques, as there are benefits to additive manufacturing that conventional manufacturing cannot provide. Using a framework can be helpful when deciding when, where, and for which instances AM could be used to create more valuable products.
During the past five years, arguably every industry has significantly grown its use of additive manufacturing (AM). Industries including aerospace, medical, and automotive, have embraced AM, also known as 3D Printing, and leveraged its inherent capabilities to accelerate the design of breakthrough products and bring those innovations to market faster.
In the aerospace industry, GE is nearing the production of flight-tested fuel injection nozzles for the LEAP engine. Before AM was used, the fuel injector was made of 20 separate components. Now, leveraging the complex geometries possible using the selective laser melting (SLM) process, GE has been able to design a single-piece nozzle that is 25% lighter and five times stronger than the previous multi-part design. Siemens has adopted AM to make burner tips for use as replacement parts in gas turbines. As a result, the time required for the repair process of burner tips has been reduced by approximately 90%.
Other industries are beginning to adopt AM for product development in very meaningful ways. For example, when the Nike team was conducting discovery interviews with football athletes they learned that most of them made adjustments to their speed and direction to compensate for how their cleats performed on the turf. For example, athletes typically reduced their speed when they came upon a turn because they feared they would overrun it, due to lack of proper traction if they were going too fast. Designing a cleat geometry to optimize performance is complex and requires a great deal of engineering and testing. To accelerate the innovation process, the Nike team used selective laser sintering to create complicated components in a matter of hours. Using AM allowed the team to create multiple design variations which in turn enabled the athletes to test multiple cleats back to back. This rapid iteration and testing allows Nike to make bigger leaps in the benefits they provide their athletes.
READ MORE ADDITIVE MANUFACTURING ARTICLES ON DESIGN NEWS:
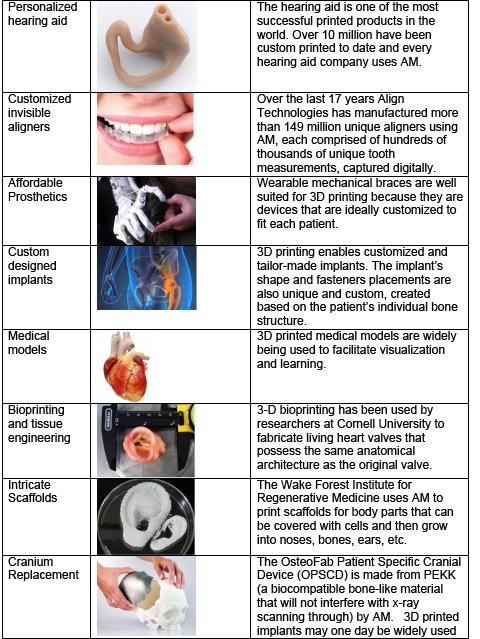
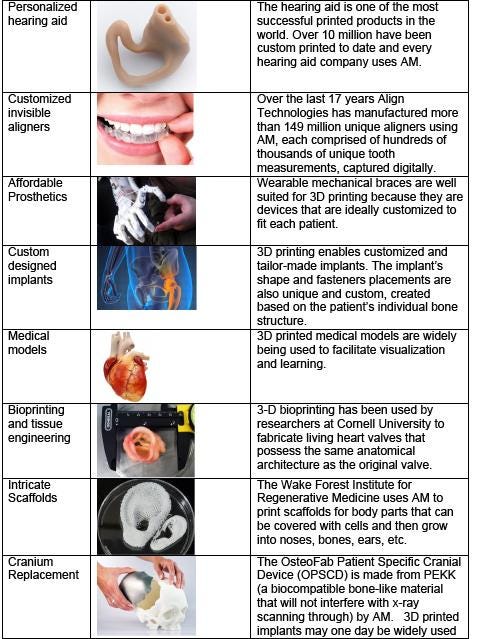
There are many other examples of how AM is being used to innovate new products, accelerate time to market, and develop personalized products that were not possible before using conventional manufacturing. It is often said AM will revolutionize manufacturing and enable the fourth industrial revolution. However, the immediate potential AM can have in medicine is less widely known and appreciated. Personalized body parts and prosthetics can improve recovery, functionality, cosmetics, and reduce procedure time and cost. From surgical implants and prosthetics, AM is transforming the field of medicine, allowing health care practitioners to create customized, patient-specific implants. Researchers are also using specialized 3D printers to grow tissue and human organs and new materials are being developed that will help rebuild tissue around 3D-printed scaffolds. The table above lists several examples including mass produced customized and personalized devices such as hearing aids and teeth aligners to intricate scaffolds for growing new body parts such as ears and noses.
Dimensions to consider when deciding between conventional techniques and AM:
Complexity: In some cases, the ability to design and make complex structures and components produces devices that have superior performance. Some medical device designs that aim to enhance performance cannot be fabricated by machining or other conventional techniques but can be produced using AM. Where functional performance can create value (in patient lifestyle, surgical success, etc.), AM should be seriously considered.
Customization: The ability to use fully customized implants can mean the difference between a device that does or does not work. For hearing aids, the use of AM has allowed companies to shrink and personalize the device and make it nearly invisible. For hip joint implants, the ability to use AM means a perfect fit thanks to printing the exact geometry and the most compatible material.
Modularization: Some medical devices have integrated electronics and other components that cannot be printed at this time. While customization can drive performance and results in a better experience for the patient and the surgeon, it is not always feasible to fully customize a medical device. For such devices, it can be highly desirable to design modular prosthetics or surgical tools and use a hybrid manufacturing approach -- conventional for electronics and other components and additive for specific tool extensions or wearables.
Time/Timing: It is widely documented that using AM can accelerate the design and development process because the approach allows for rapid prototyping and iteration. In the medical industry, "timing" is even more important. Some situations need just-in-time custom fabrication to save lives. In these cases, 3D printing is by far the most effective way to quickly produce specific medical devices or implants.
Cost: Many medical devices are good candidates for additive manufacturing due in part to their apparent lower-volume production needs. The cost of producing medical devices is high and for certain applications not affordable for some patients. For example, the cost of a prosthetic arm can range from $5k to $50k. This is not an issue for adults who typically use the prosthetic for many years because the cost can be spread over a long period. For children however, because they are growing fast, they need a new prosthetic arm or leg every few months. Insurance companies typically cannot and do not cover the cost of a new prosthetic. Using AM can cut the cost of producing prosthetics by an order of magnitude and allow children to have a new prosthetic every few months.
In summary, designing and producing medical devices using AM opens many opportunities to optimize performance, rapid test multiple design options, obtain real-time patient and surgeon feedback, accelerate medical care, and de-risk the overall medical device innovation process. Therefore, to remain on the cutting edge, medical device companies and healthcare practitioners need to build the skill set to know which tools to use, when to use them, and which suppliers to involve in their medical device innovation effort.
John Jaddou (pictured at top) is a managing director at Veraxia and works with companies on business development, innovation management, technology commercialization, and product strategy. John is also the co-founder of Addeation, a consultancy focused on design strategy and additive manufacturing for accelerated product development. More recently he was a director at Cabot Corporation where he built the global innovation program, established the 3D printing/additive manufacturing business, and led the introduction of a new product blueprinting methodology to boost the front end of the innovation pipeline. John has been working with fortune 500 companies and helping business leaders harness the value of 3D printing/additive manufacturing and integrate digital design, development, and commercialization tools into their business model for a sustainable competitive advantage. John holds an MSc in Engineering from the University of Michigan and an MBA from Michigan Ross School of Business.
A. John Hart is associate professor of Mechanical Engineering and Mitsui Career Development Chair at the Massachusetts Institute of Technology. He has PhD and S.M. degrees from MIT, and a B.S.E. degree from Michigan, all in Mechanical Engineering. At MIT, John leads the Mechanosynthesis Group, which creates new machines, materials, and design principles for advanced manufacturing. He teaches courses in manufacturing processes, nanomaterials, and research methods, including a week-long professional program on additive manufacturing at MIT. He is also co-founder of Addeation.
Like reading Design News? Then have our content delivered to your inbox every day by registering with DesignNews.com and signing up for Design News Daily plus our other e-newsletters. Register here!

Design News will be in Minneapolis and Orlando in November! Design & Manufacturing Minneapolis will take place Nov. 4-5, while Design & Manufacturing South will be in Orlando Nov. 18-19. Get up close with the latest design and manufacturing technologies, meet qualified suppliers for your applications, and expand your network. Learn from experts at educational conferences and specialty events. Register today for our premier industry showcases in Minneapolis and Orlando.
About the Author(s)
You May Also Like





