Using Magnets as a Depression Treatment
October 11, 2010
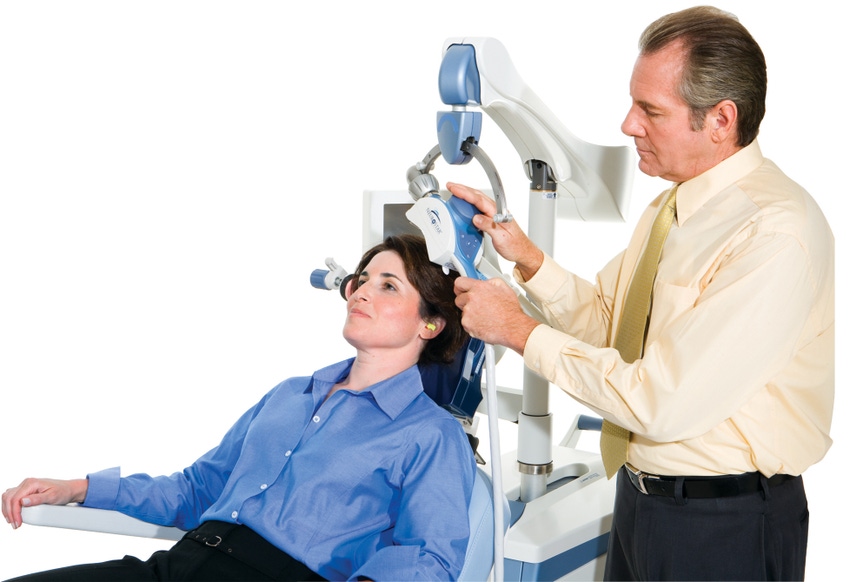
Majordepression is a serious medical disorder affecting more than 14 millionAmerican adults every year. Often a debilitating condition, depressionresults in a persistent state of sadness which interferes with an individual'sthoughts, behavior, mood and physical health.
Commontreatment for sufferers involves pharmaceutical products prescribed byphysicians that are sometimes required for extended periods of time. Medications and professional counseling willhelp usually help patients manage the negative effects of depression but forsome, anti-depressants fail to provide improvement. Extreme cases can result in completeincapacitation of the patient and hospitalization.
TMS.NeurostarPatient |
As analternative to the pharmaceutical approach to depression treatment, NeuroneticsCorporation of Malvern, PA, has designed the NeuroStar TMS (transcranial magneticstimulation) Therapy System. TMS is a non-invasive, non-systemic treatment fordepression that applies a highly focused, pulsed magnetic field to stimulatefunction in targeted brain regions. NeuroStar TMS Therapy is performed in aphysician's office. Each treatment lasts about 40 minutes and takes place dailyfor four to six weeks. To date,Neurostar reports that there are have been none of the physical effects reportedthat are commonplace with drug treatment. Patients can immediately return tonormal daily activities.
Designing the TMS Device
Neurostar engineersdeveloping the TMS treatment needed a reliable means of holding the treatmentcoil in place during therapy sessions and turned to Ogura IndustrialCorporation for suggestions. This was a major design consideration as it isnecessary for the magnetic treatment coil to be able to be positioned indifferent axes of motion to accommodate the different physical characteristicsof individual patients.
Once thepatient is reclined in the treatment chair, the treatment coil is positioned sothat magnetic pulses are focused on specific regions of the brain. It is critical that, once the coil ispositioned and therapy commences, no slipping or wandering of focus location occurs. The magnetic pulses generated by the treatmentcoil must remain on the targeted area of the brain.
TMS.RNBBrake |
RNB electromagneticspring-applied brakes from Ogura were selected to hold the mast and gantry arm ofthe TMS securely during the therapy sessions. These brakes, designed forholding and emergency braking, are considered power-off brakes in that no poweris needed to hold the brake, due the use of a spring to keep it in place.
This was animportant consideration in the TMS design, since quiet operation is essentialin a therapeutic environment. Since the RNB power-off brakes are only energizedwhen the treatment coil is being positioned, the treatment coil, once put intoplace, is held securely in place with no electrical noise or "hum" to distractthe patient.
Because theRNB is a power-off brake, there is also no electrical signal to interfere withthe TMS system during use. Power is onlyapplied to release the brake when the treatment coil is adjusted to theindividual patient. When power isremoved, springs apply the friction surfaces locking the mast and gantry arm inplace until the therapy session is completed. In addition, the zero backlash feature of theRNB design eliminated the possibility of flutter or drift of the gantry arm andmast during operation.
Although itwas not a primary design consideration, cost of operation of the RNB isnegligible. Power consumption is minimized since the RNB is only energizedwhile adjusting the system to the individual patient. During treatment the RNB is static, stable, non-interferingand power-free.
About the Author(s)
You May Also Like





.jpg?width=300&auto=webp&quality=80&disable=upscale)

