June 24, 2011

As medical devices become smaller and more mobile, motion control solutions have been keeping pace by delivering new innovations, smaller footprint packages, and reduced power needs. But the key to these new solutions for miniature motion control is a much higher level of integration of the motion subsystem into the medical device itself.
Use of integrated motor drivers and modular controllers provides design tools that reduce the hardware required to implement control systems. New human-scale fluid power devices are combining ultra-low-power consumption valves, force, and position sensors with control electronics to create new solutions such as an untethered ankle foot orthosis.
Along with a continuing focus on performance, the design challenge is to achieve the ultimate in reliability while making motion components more compact and intelligent.
Design trend to integrated solutions
"For each segment within the medical device space, motion control applications face different concerns, but the common theme across them all would be reliability, controllability, and more advanced control using feedback and closed-loop systems," says Ryan Kehr, applications manager, motor products, for Texas Instruments. "Especially with the certifications required in the medical space, it is important that these things are proven out."
Kehr says there are three basic motor types used in medical devices:
Brushed dc motors operate in an on/off mode suitable for simpler applications that require lower precision.
Stepper motors provide higher precision where there is a requirement to count the exact number of milligrams that need to be dispensed.
Complex three-phase brushless solutions provide the highest level of reliability, but involve more complicated controls for applications like ventilators or cooling fans in medical instrumentation devices.
"A specific area we're seeing as a design trend is integrated motor drivers," says Chris Griffith, medical business development manager for Texas Instruments. "In the past, many of these applications have used discrete solutions where there are separate gate drivers and MOSFETs, and it is often difficult to provide the type of protection that an integrated solution can offer. Robustness for medical devices is a key advantage that we can provide by integrating the solution."

By integrating components into a single package with on-chip over-current, over-temperature, and shoot-through protection, there is an ability to make sure the high- and low-side FETs are not turned on at the same time, which can cause device failure. The approach also addresses cost from a PCB area standpoint, and shrinking board sizes tend to reduce the overall size of the end product. The reduction in board space can be up to 50 percent by using an integrated versus a discrete solution.
"Many of the products in our portfolio can be used for both stepper and dc brushed control," says Kehr. "Some have an onboard microstepping capability, which minimizes the amount of external controls needed for step and direction applications, for example. For a urine analyzer moving between different vials, a stepper can provide a good positioning solution. For a fan motor in a ventilator spinning at a high RPM, a brushless dc device fits well and provides performance gains, which can provide increased efficiency in a smaller form factor. The long-term reliability of brushless is also a plus for applications such as ventilators."
He says ongoing work with brushless dc systems is focusing on sensorless control algorithms. These systems can provide a cost advantage and eliminate another area where you can have a failure mechanism using external sensors like Hall Effect sensors required for sensored brushless dc control.
In sensorless applications, feedback is provided by measuring back EMF voltages and phase currents. This requires a high slew rate, especially for applications such as fan motors, which are spinning at a higher speed; precise feedback on the currents and back EMF voltages; and often implementation of signal conditioning using op amps. The technical challenge is to make sure the current is calibrated from the standpoint of offsets to prevent introducing commutation errors.
Human-scale fluid power devices
The development of a light, compact, efficient, ultra-low-powered, untethered Ankle Foot Orthosis (AFO) system has the potential to yield significant advances in orthotic control mechanisms and treatment strategies.
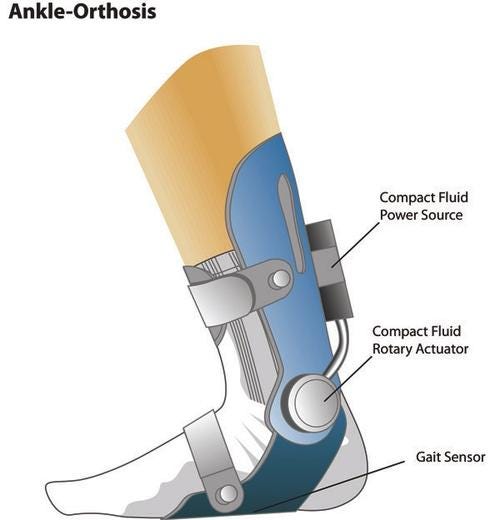
Active control of the joint, versus an orthosis which functions as a brace, is achieved by offering a truly mechatronic-style set of components. The new orthosis provides both the necessary propulsion and an ability to adjust foot position during the gait for patients suffering from medical conditions and injuries. In collaboration with a leading research institution, Parker Hannifin is developing a motion control solution for this new category of human-scale fluid power devices.
"The core piece of the innovation is the optimization and development of a dual vane rotary actuator that is high torque and miniature in size," says Richard McDonnell, product sales manager for Parker Hannifin Corp.'s Automation Group. "A rotary actuator at the apex of the ankle bone allows the motion using compressed air, and a piezo valve controls the bi-directional movement of the actuator. Individual sensors at the toe and heel provide feedback on the position of the foot, and are used by the actuator to provide assistive power and propulsion."
Enabling technologies for the system are the advent of ultra-low-power consumption valves, force and position sensors, and control electronics. McDonnell says the key is the ability to take a traditional rotary actuator and lighten it up to offer minimal weight for the wearer, making it compact while providing a substantial amount of shock and vibration protection. Use of pneumatics offers a lightweight solution that also delivers the desired motion profile.
A core challenge with the non-tethered AFO is the need it creates for a compact power source, which requires a pneumatic reservoir and becomes another component the patient will carry. The reservoir needs to be compact but also provide high enough pressure to sustain several thousand steps of use for the patient.
Other challenges include combining the compactness and efficiency of the actuators with the effective transmission of pneumatic power to provide the desired torque from the actuator. Component integration for reduced size and weight is a key design constraint, along with smart control schemes that accomplish functional tasks during gait and effectively manage the human machine interface (HMI).
"In the US alone, there are more than 6.2 million individuals who would benefit from a portable, powered, daily wear AFO. For individuals with impaired ankle function, current solutions are passive braces that provide only motion control and joint stability," says McDonnell. "These designs often fail to restore normal ankle function because they lack the ability to actively modulate motion control during gait, and cannot produce propulsion torque and power."
Next Page: Ventilators & controllers
About the Author(s)
You May Also Like





